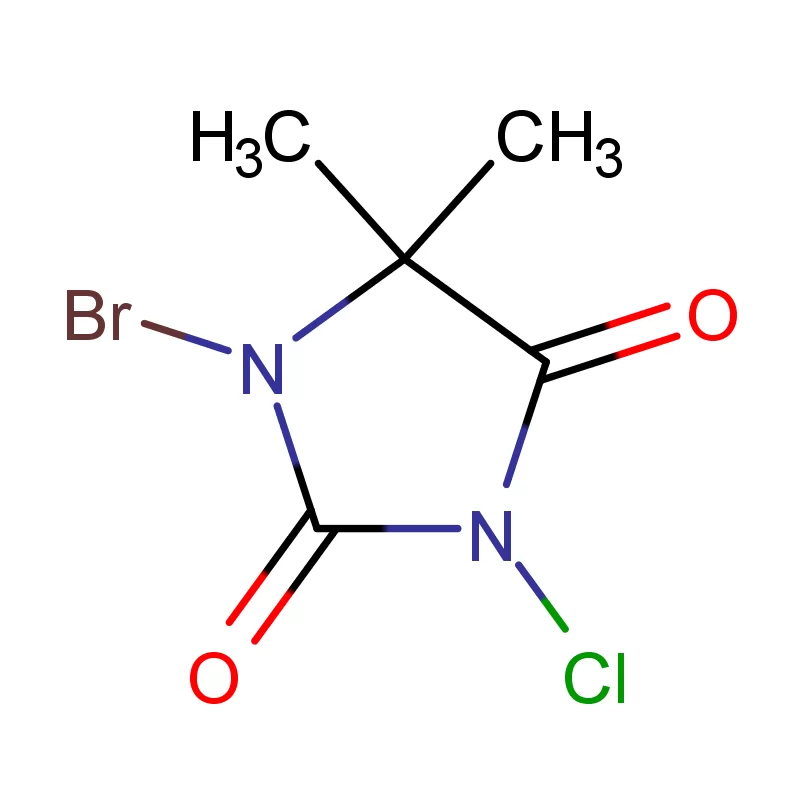
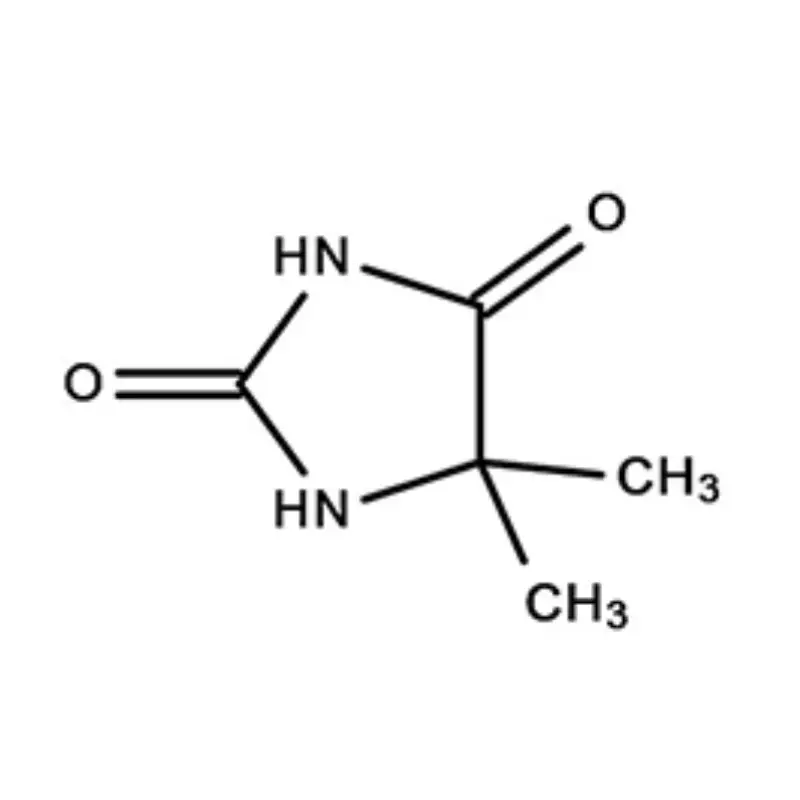
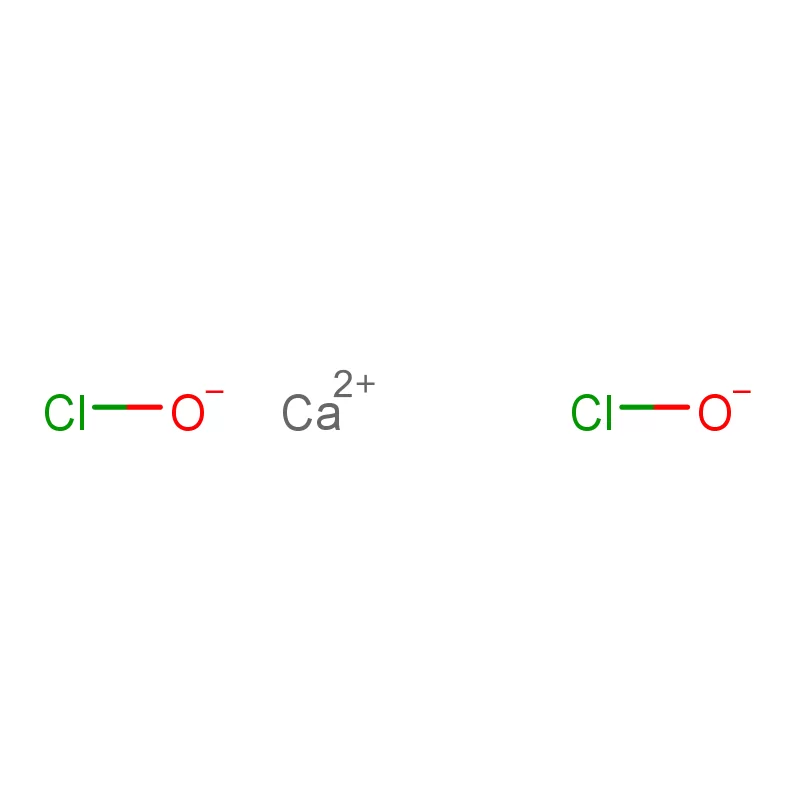
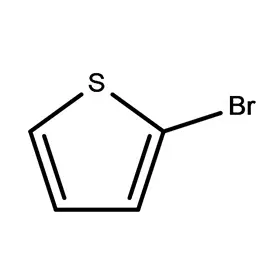
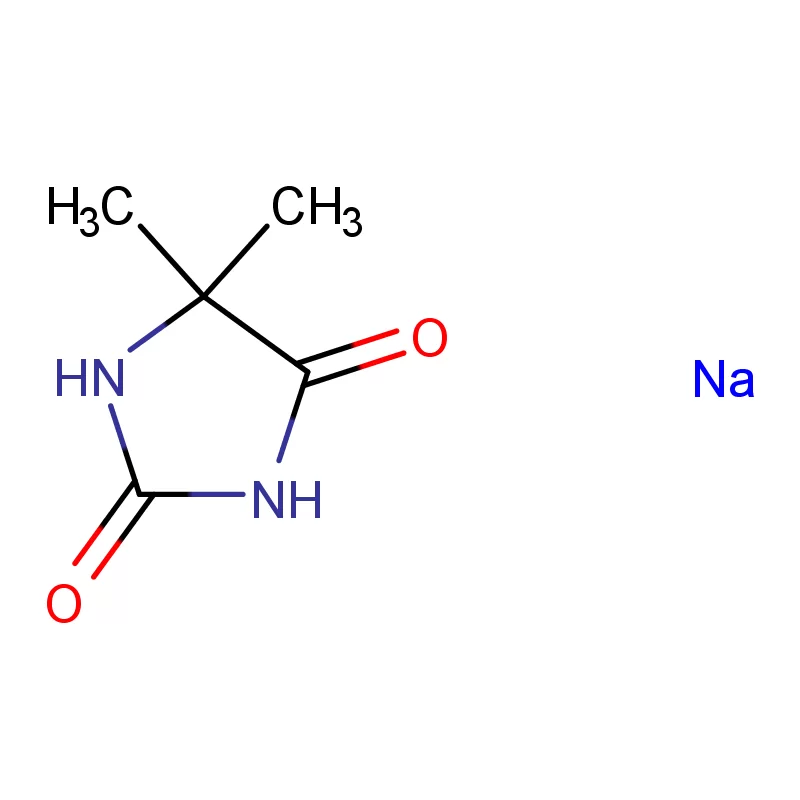
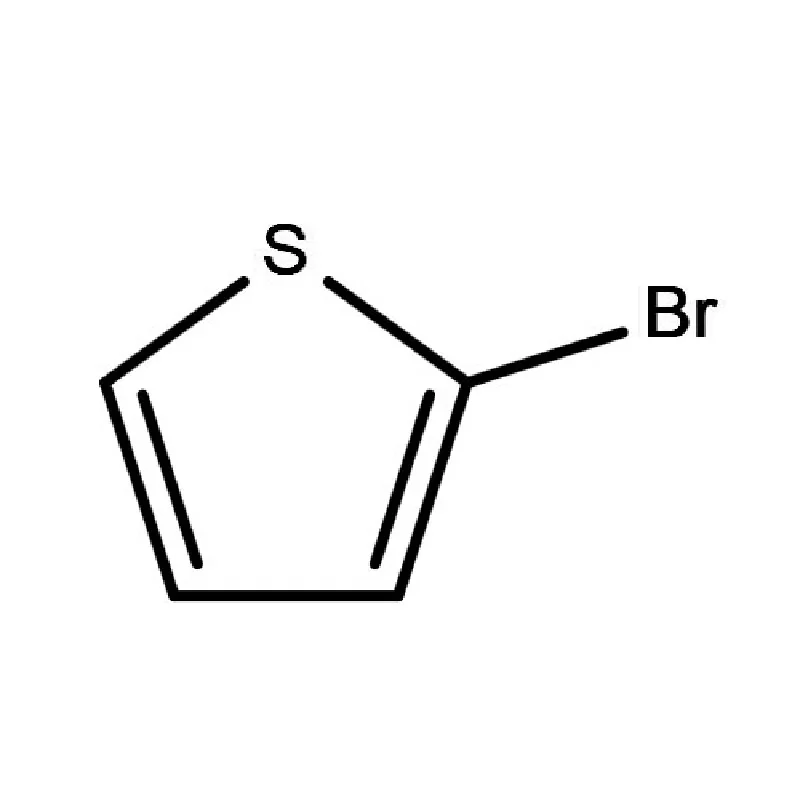
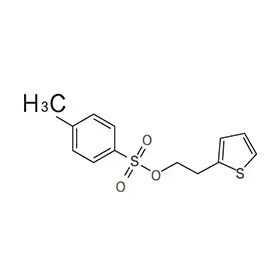
Ghana's pharmaceutical sector is experiencing significant growth, driven by increasing healthcare access and a growing population. However, the reliance on imported pharmaceutical intermediates remains a key challenge. Local manufacturing capacity is developing but often hindered by access to high-quality raw materials and specialized fine chemicals.
The climate of Ghana, being tropical, presents unique storage and transportation considerations for sensitive chemical compounds. Maintaining the integrity of 2 Bromothiophene and other temperature-sensitive materials requires robust cold chain logistics. Economic factors, including currency fluctuations and import duties, also impact the cost and availability of raw materials.
Government initiatives are aimed at strengthening the local pharmaceutical industry, including promoting local production and attracting foreign investment. This creates opportunities for suppliers of quality industrial water treatment chemicals and pharmaceutical raw materials.