Austria's pharmaceutical sector is a key contributor to the nation's economy, representing a significant portion of its manufacturing output. The demand for high-quality pharmaceutical intermediates and fine chemicals is consistently strong, driven by both domestic production and export requirements. Austria benefits from a highly skilled workforce and stringent quality control regulations.
Geographically, Austria’s central European location provides advantageous logistics for distribution across Europe. The climate, while temperate, necessitates careful control of storage and transportation conditions for sensitive chemical compounds. Furthermore, Austria's commitment to sustainable manufacturing practices is increasing the demand for environmentally friendly chemical processes.
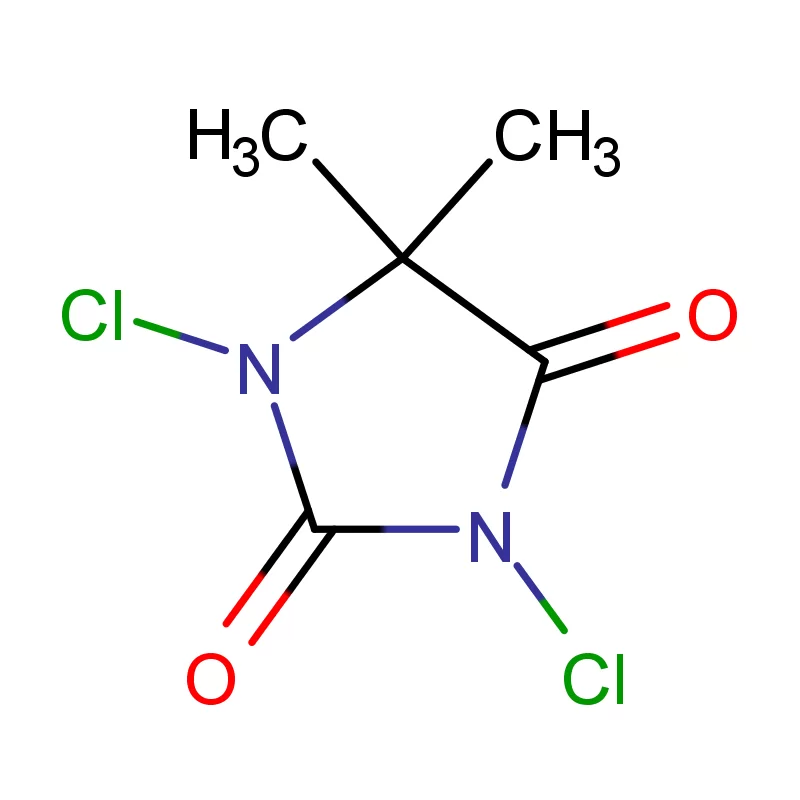
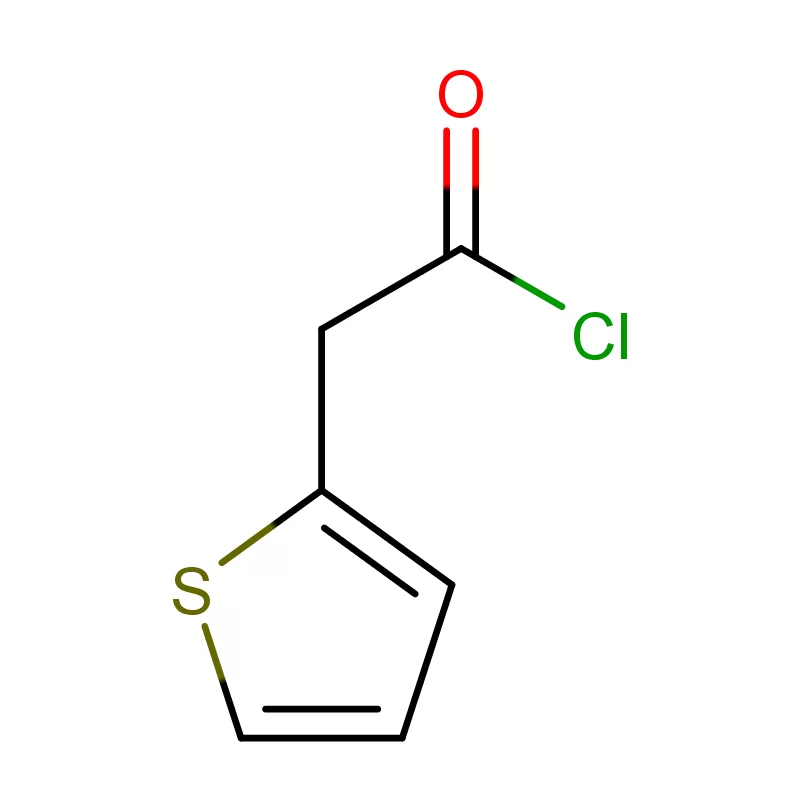
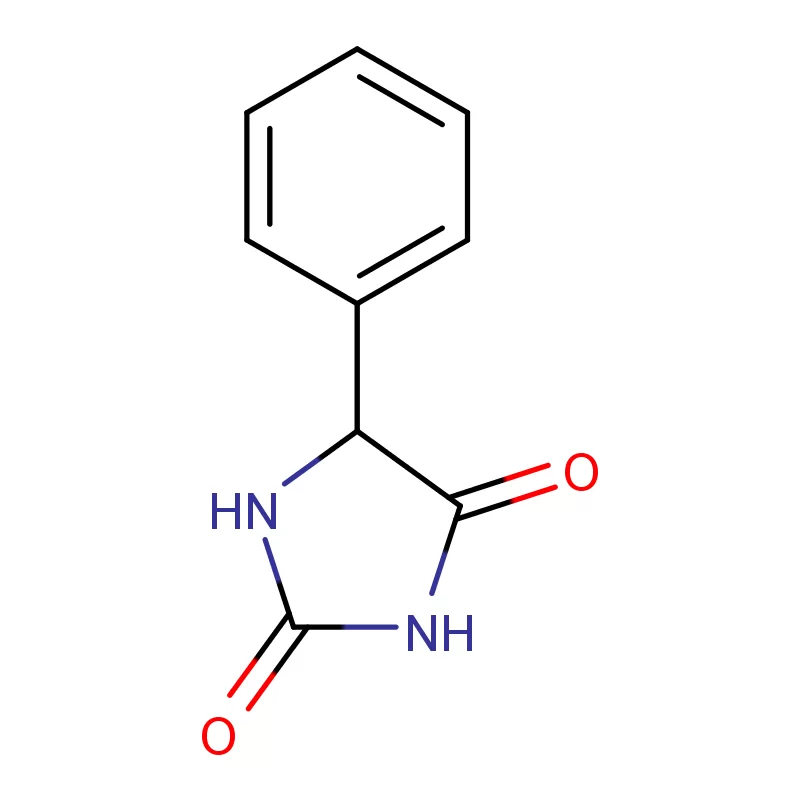
The Austrian market, while relatively small compared to larger European economies, represents a high-value segment for specialized chemical suppliers. The focus is heavily on innovation and advanced manufacturing techniques. Manufacturers are seeking reliable partners who can provide consistent quality and adherence to strict regulatory standards regarding 2 Bromothiophene and other specialized compounds.