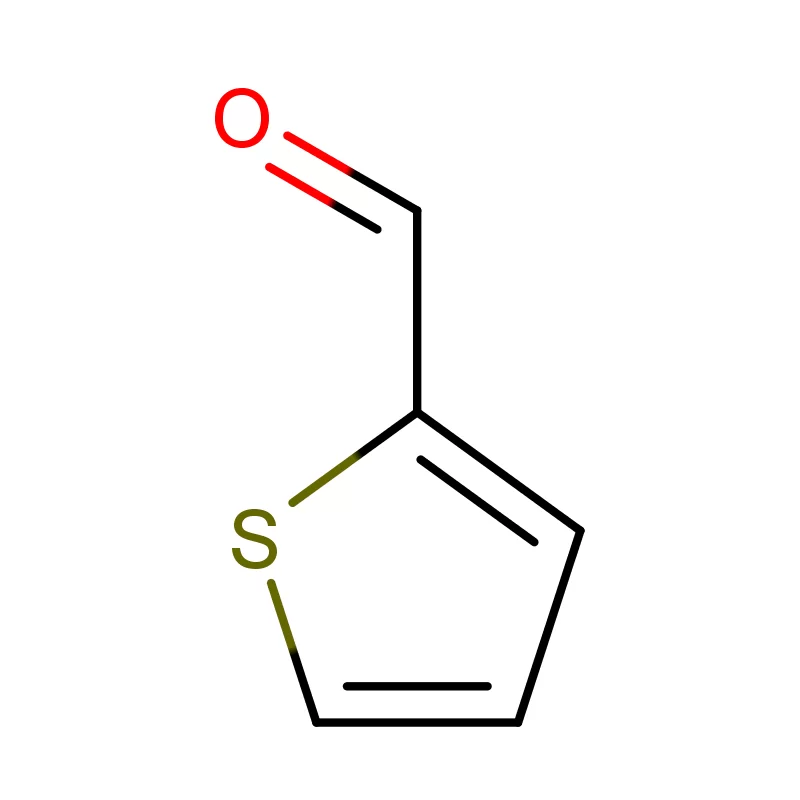
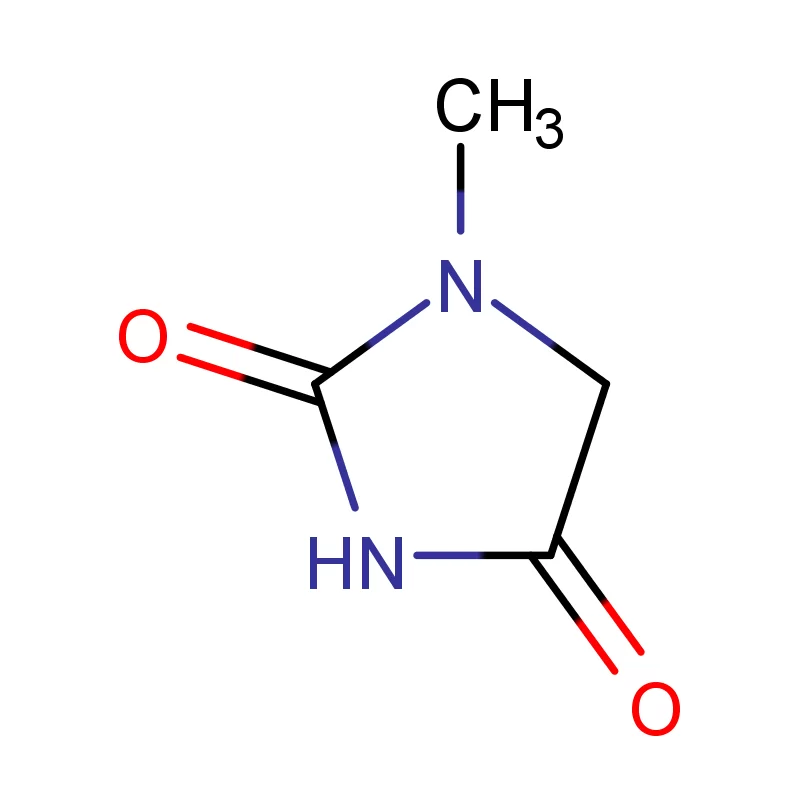
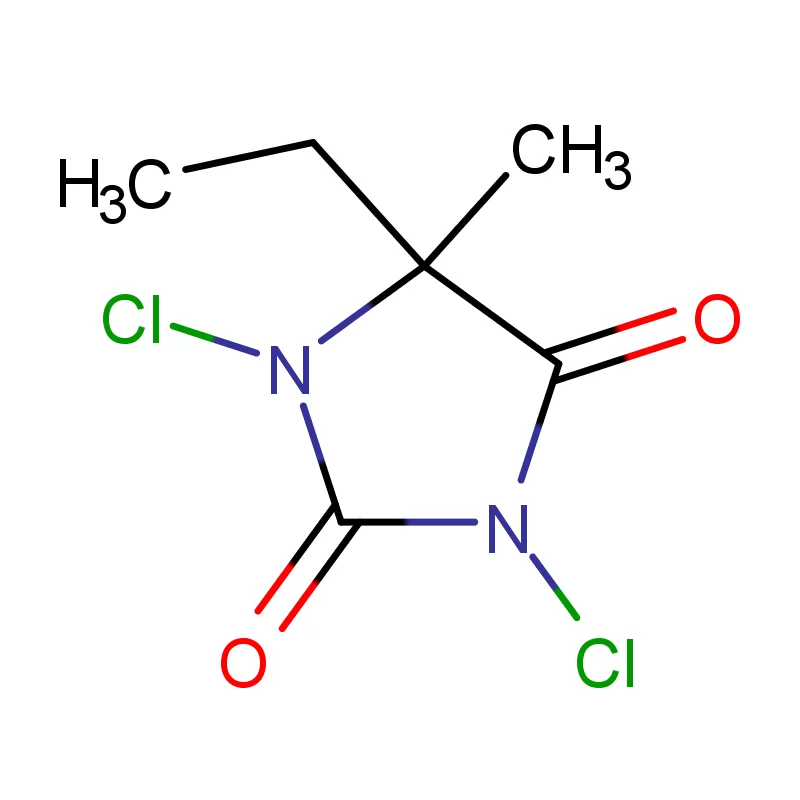
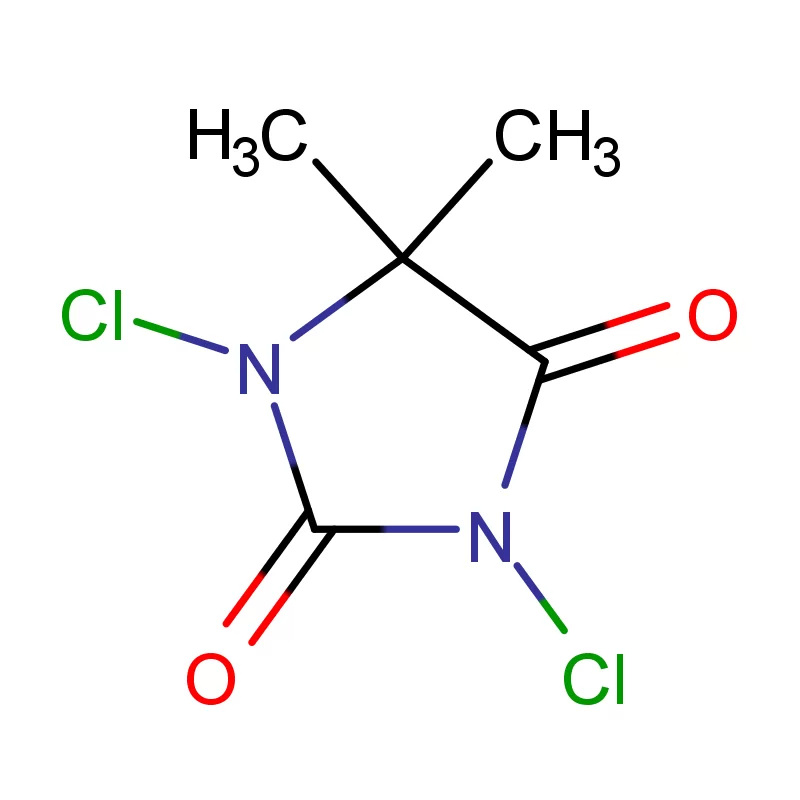
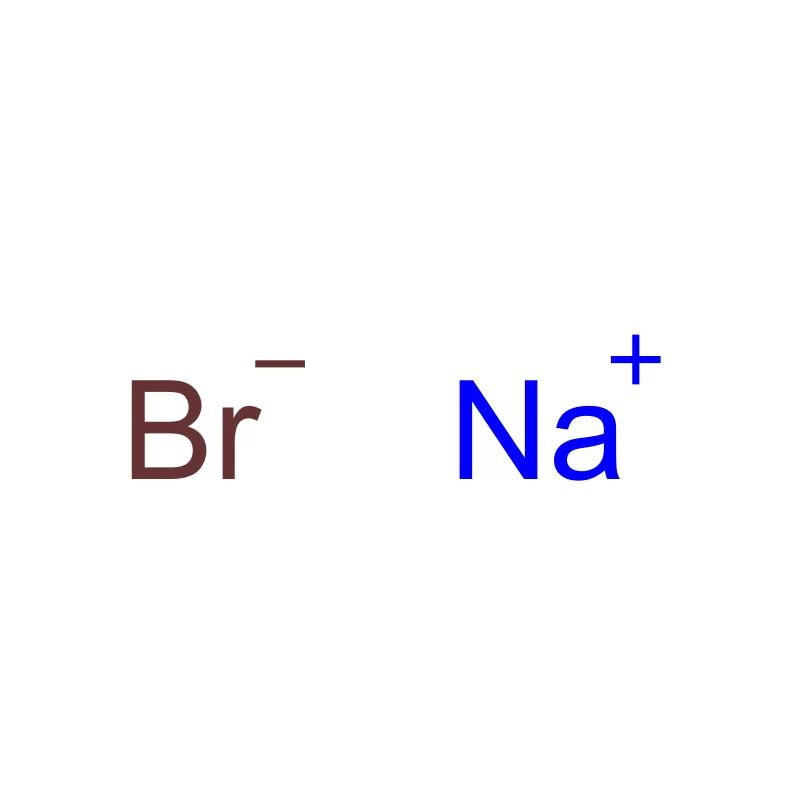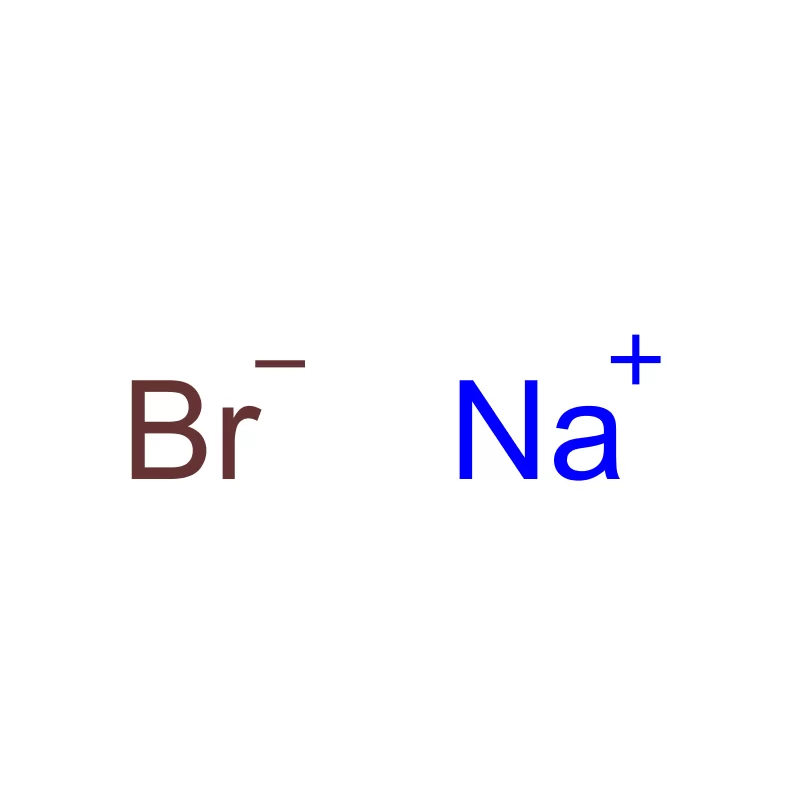Indonesia, as the largest economy in Southeast Asia, presents a significant and rapidly expanding market for pharmaceutical intermediates. The country's growing population, increasing healthcare awareness, and rising disposable incomes are driving demand for pharmaceutical products, consequently increasing the need for high-quality chemical inputs. The archipelago’s geography presents logistical challenges, but also opportunities for localized production and supply chains.
The Indonesian pharmaceutical industry is currently experiencing a shift towards greater self-sufficiency, with the government actively promoting local manufacturing of Active Pharmaceutical Ingredients (APIs) and intermediates. This is partially driven by a desire to reduce reliance on imports and strengthen national health security. However, this requires substantial investment in technology and skilled labor.
Competition within the industry is intensifying, with both domestic and international players vying for market share. Regulatory compliance, particularly with international standards like cGMP, is crucial for success. Industrial water treatment chemicals are becoming increasingly important as manufacturers focus on sustainable and environmentally responsible practices.