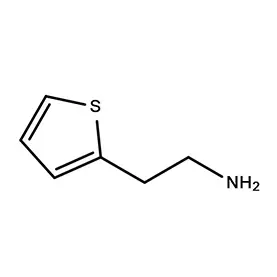
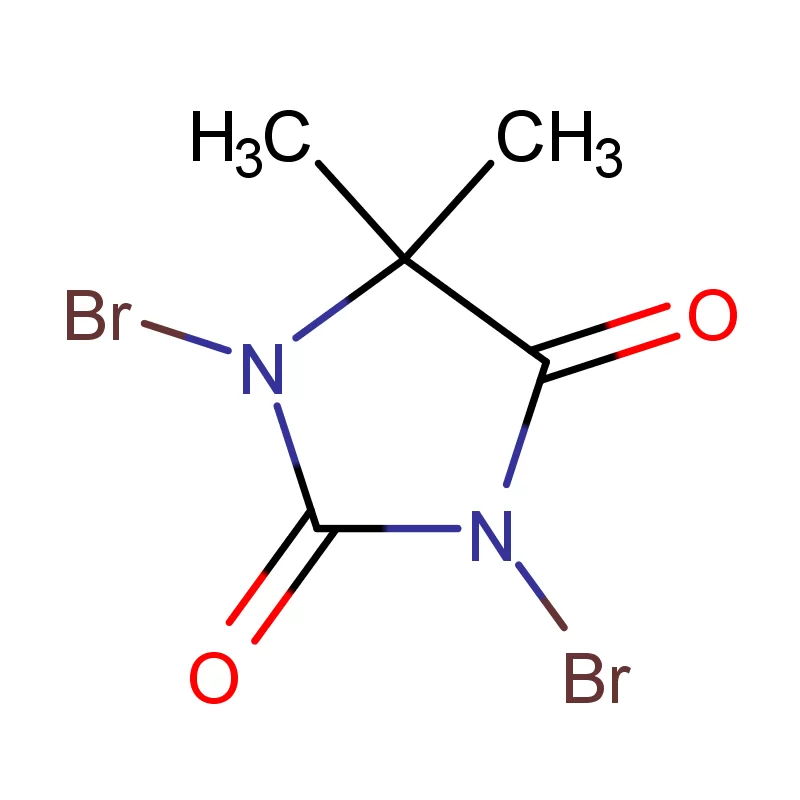
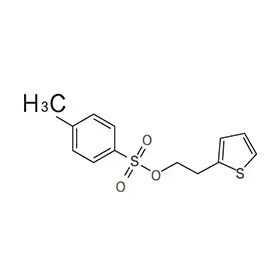
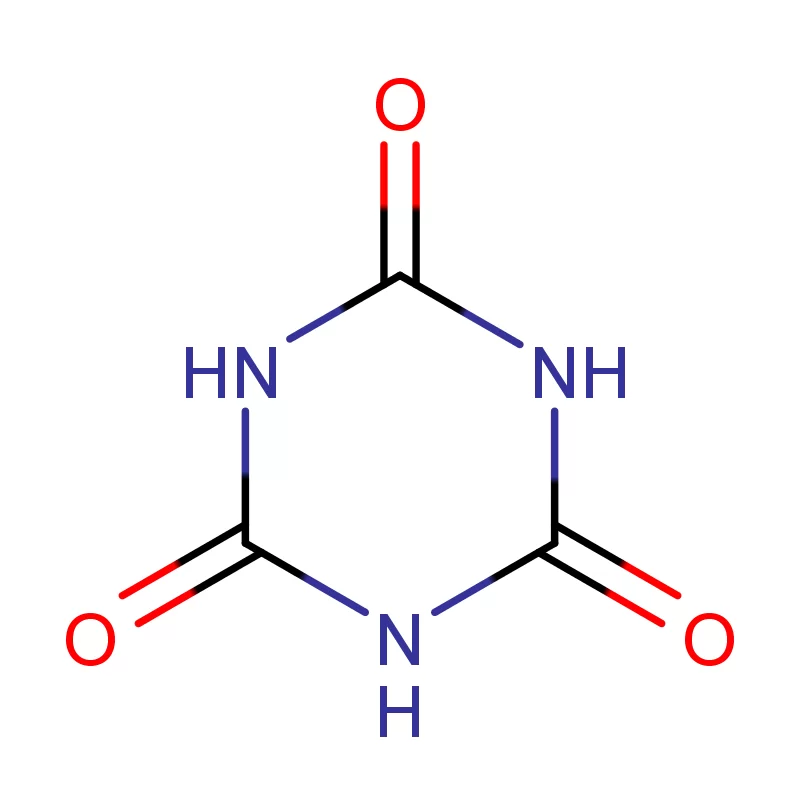
Morocco's pharmaceutical sector is experiencing growth, driven by increased healthcare spending and a rising population. The demand for pharmaceutical intermediates is consistently increasing, as local manufacturers seek to reduce reliance on imports. Geographically, the concentration of pharmaceutical production is largely centered around Casablanca and Tangier, benefitting from established infrastructure and proximity to ports.
The climate of Morocco, characterized by hot, dry summers and mild, wet winters, necessitates specialized storage and transportation solutions for sensitive fine chemicals. This presents both challenges and opportunities for companies offering robust supply chain management and temperature-controlled logistics. Economic factors, including government initiatives to promote local production, are also shaping the industry.
Furthermore, Morocco’s growing focus on sustainable development is boosting demand for environmentally friendly industrial water treatment chemicals. Pharmaceutical manufacturers are increasingly adopting responsible waste management practices, creating a market for specialized treatment solutions.