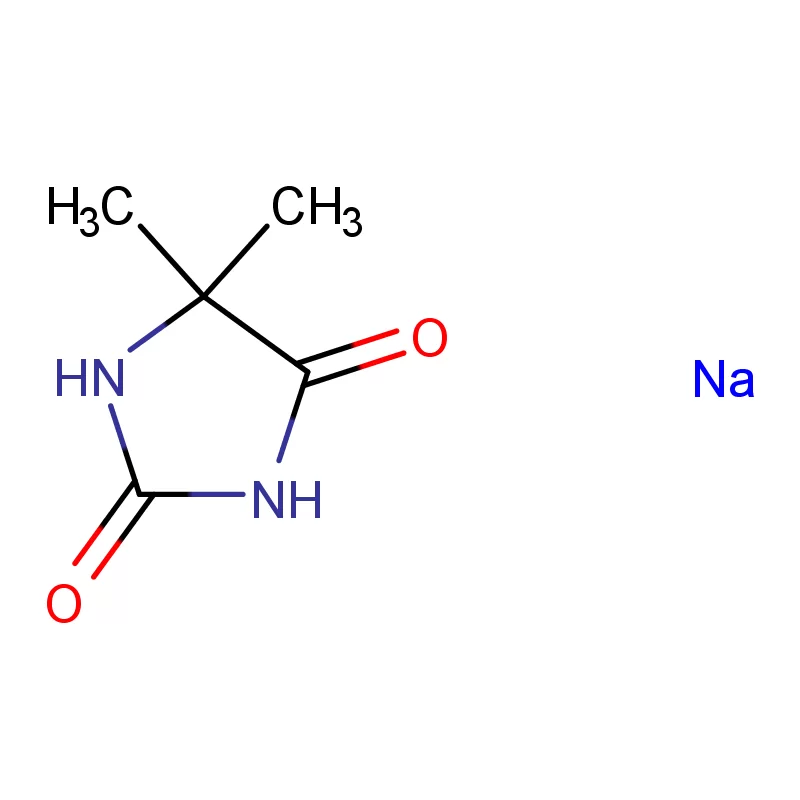
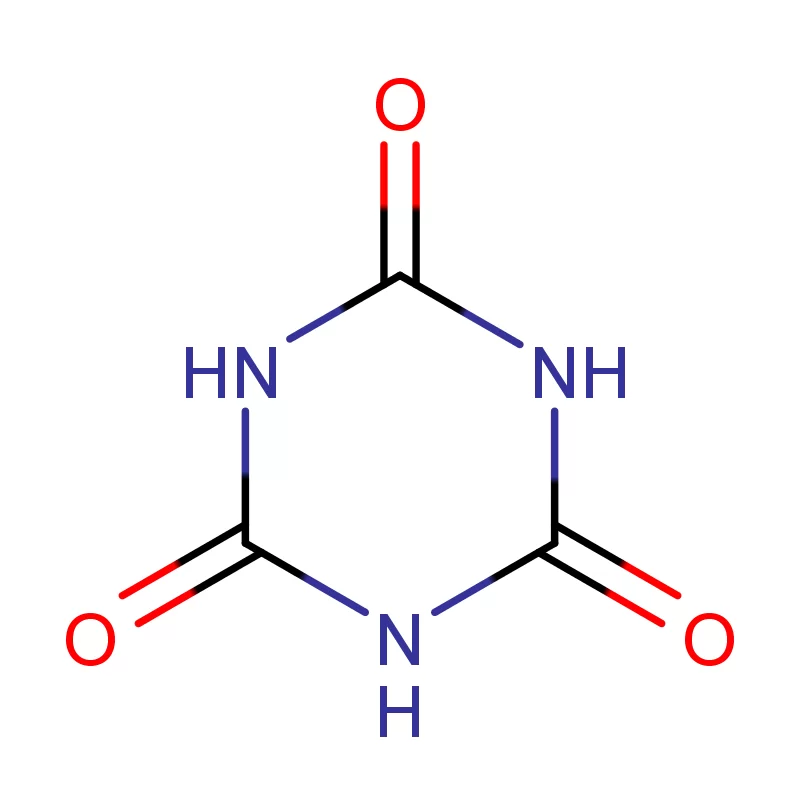
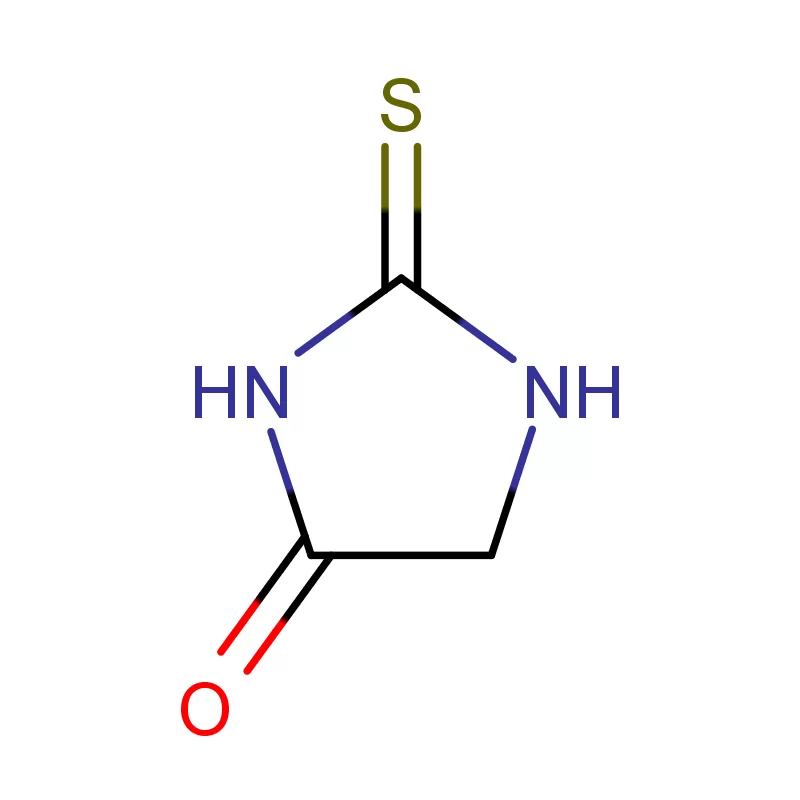
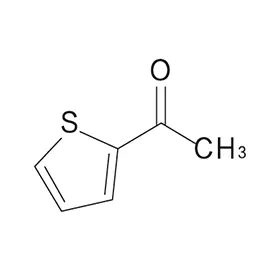
The pharmaceutical industry in Europe and Russia presents a complex landscape shaped by stringent regulatory requirements, increasing demand for generic drugs, and a growing focus on innovation. Europe, with its well-established pharmaceutical manufacturing base, demands high-quality pharmaceutical intermediates and consistently reliable supply chains. Russia, meanwhile, is increasingly focused on import substitution, fostering a demand for locally sourced or readily available chemical building blocks.
Geopolitical factors and economic fluctuations significantly impact the import and export of fine chemicals. Sanctions and trade barriers can disrupt supply chains, creating opportunities for alternative sourcing strategies and bolstering domestic production capabilities. The climate also influences logistical considerations, particularly in Russia's vast territories.
Furthermore, the drive for sustainability and green chemistry is influencing the demand for more environmentally friendly manufacturing processes and sourcing of 2 Bromothiophene and other key intermediates. Companies are increasingly seeking suppliers committed to responsible chemical management and reducing their environmental footprint.