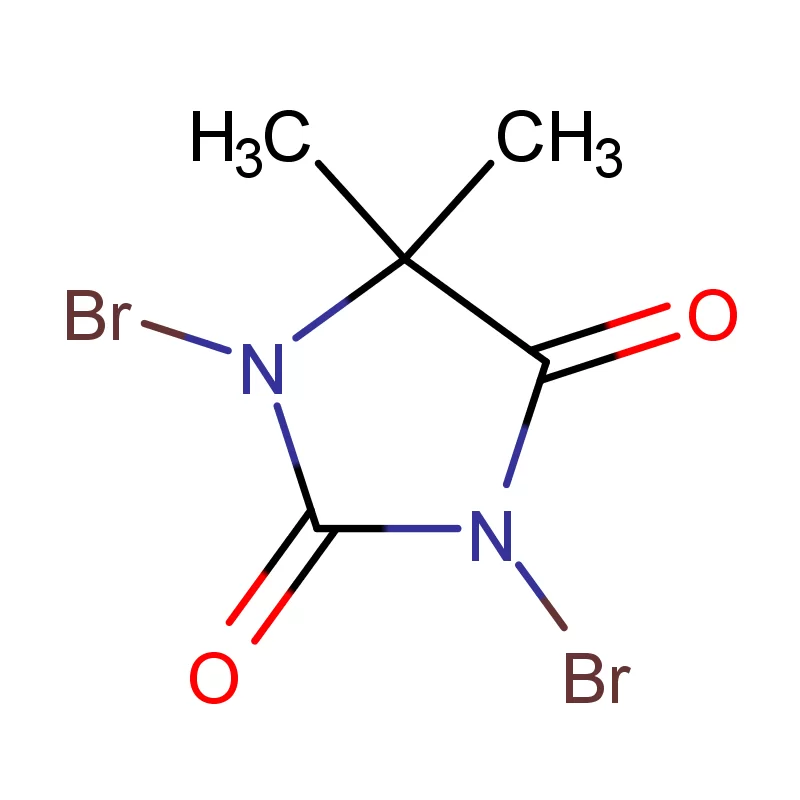
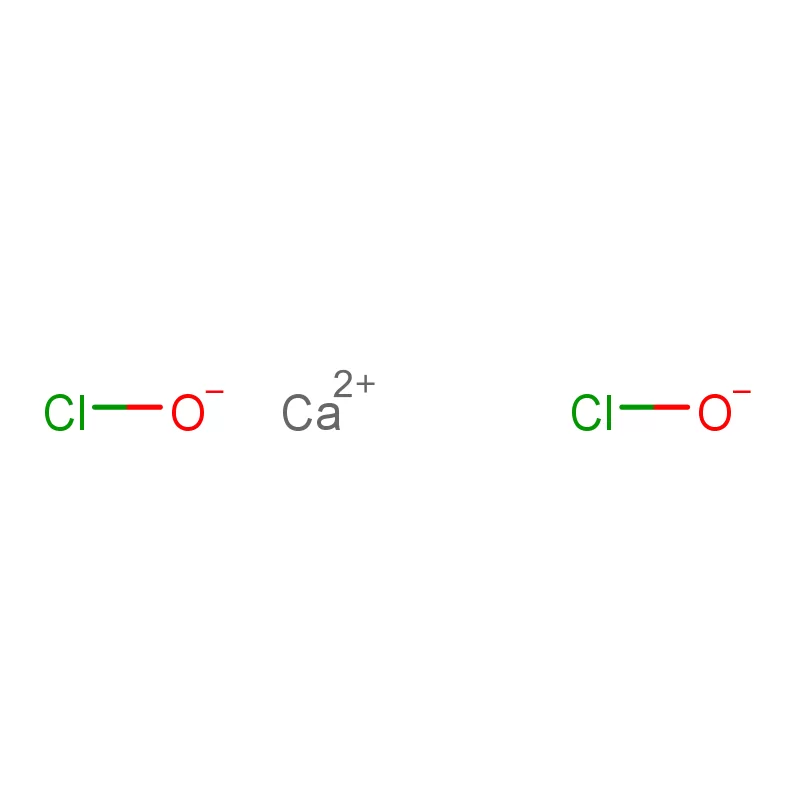
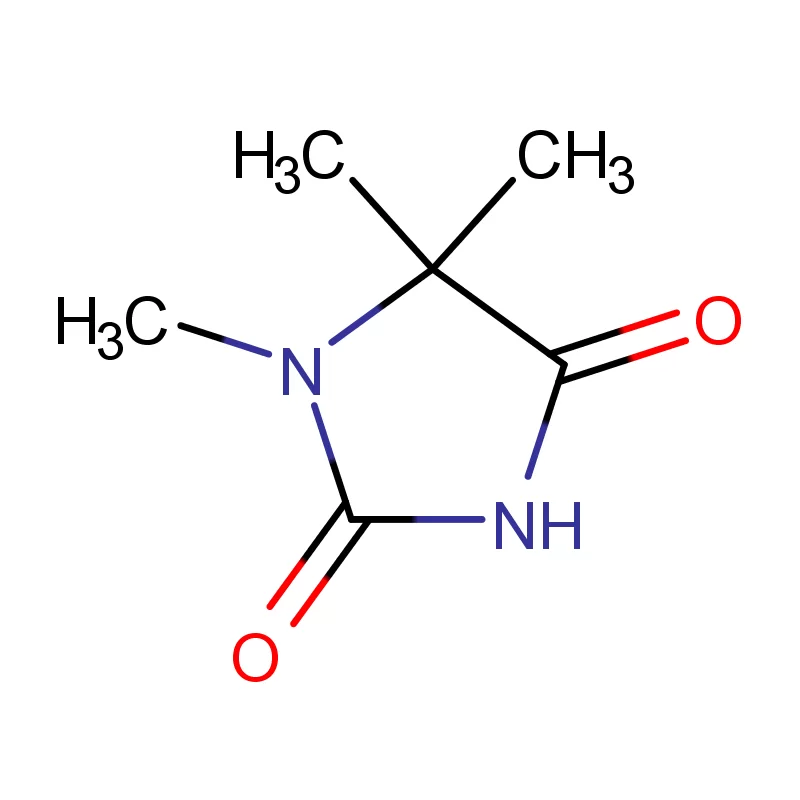
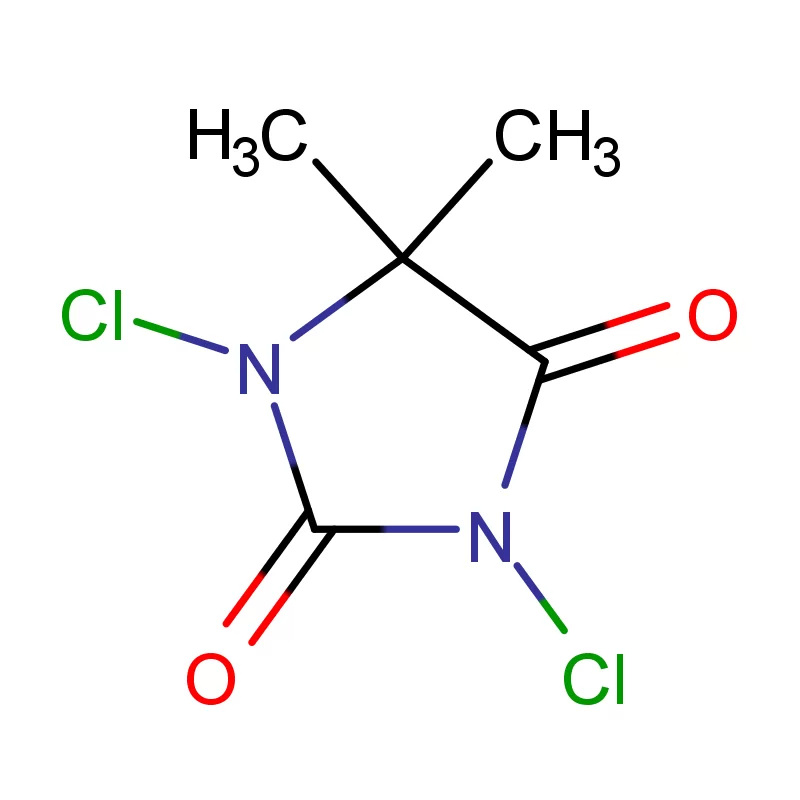
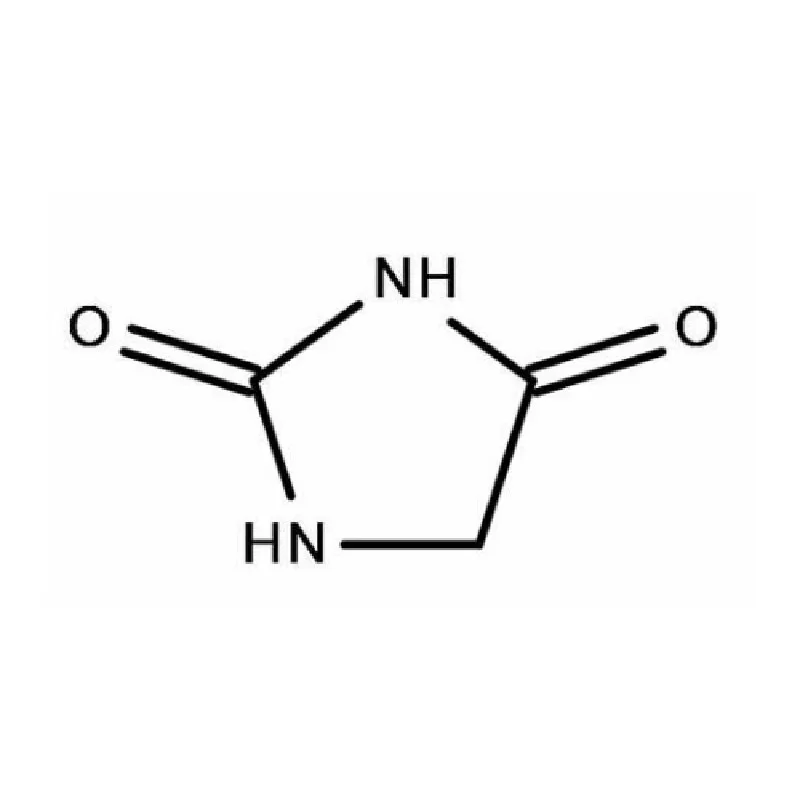
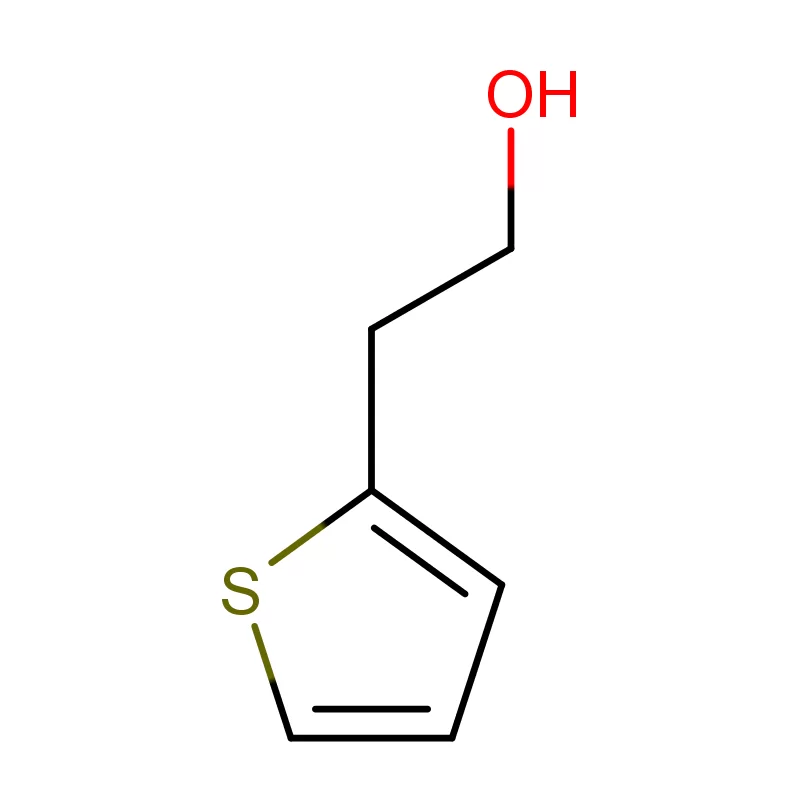
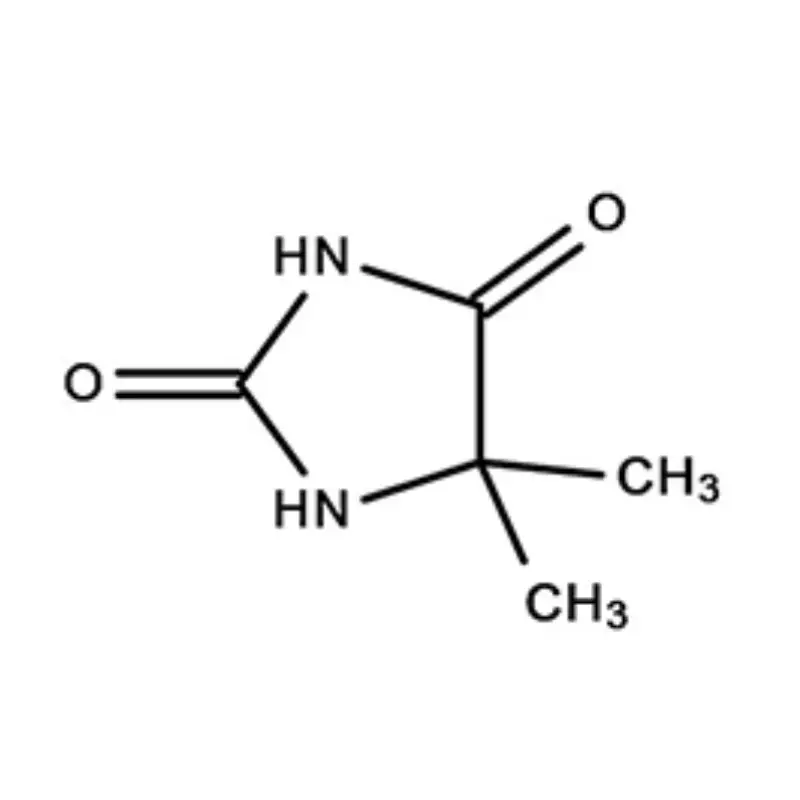
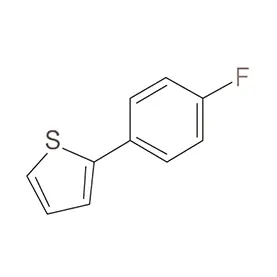
Laos, as a developing nation in Southeast Asia, has a growing but relatively nascent pharmaceutical industry. The sector is heavily reliant on imports of both finished pharmaceutical products and pharmaceutical intermediates. Geographical factors, including a landlocked position and mountainous terrain, contribute to logistical challenges and higher import costs. This creates a demand for locally sourced materials and streamlined supply chains.
The climate in Laos, characterized by high humidity and distinct wet and dry seasons, impacts storage and handling requirements for sensitive fine chemicals. This necessitates robust quality control measures and specialized infrastructure. Furthermore, economic factors, such as limited local investment and reliance on foreign aid, influence the pace of industry development.
Despite these challenges, Laos is actively working to strengthen its pharmaceutical manufacturing capabilities through government initiatives and collaborations with international partners. There’s increasing focus on generic drug production and improving access to essential medicines for its population. The market shows potential for growth, especially with an expanding healthcare infrastructure.