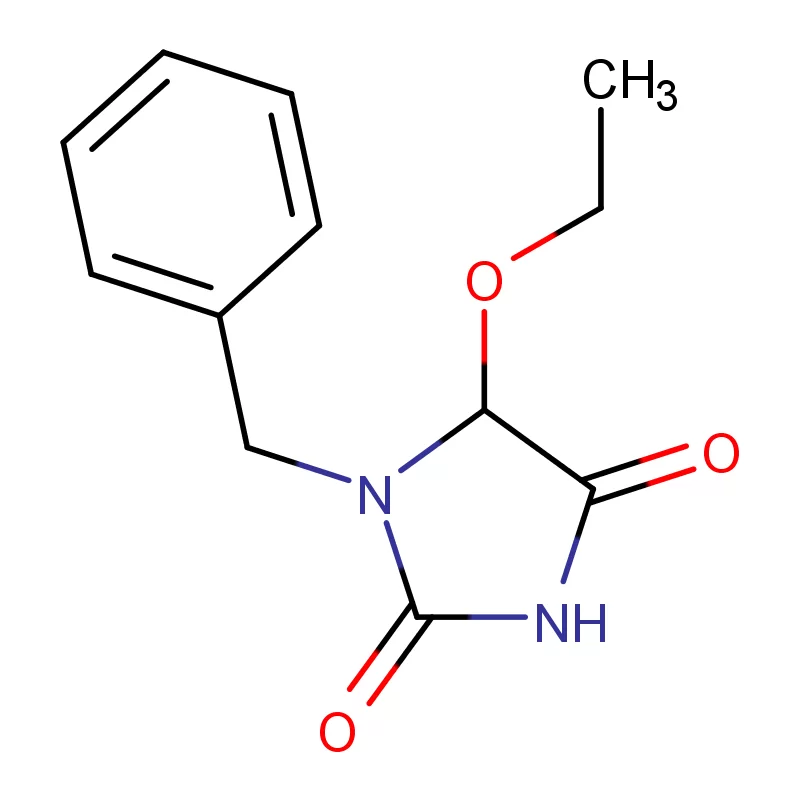
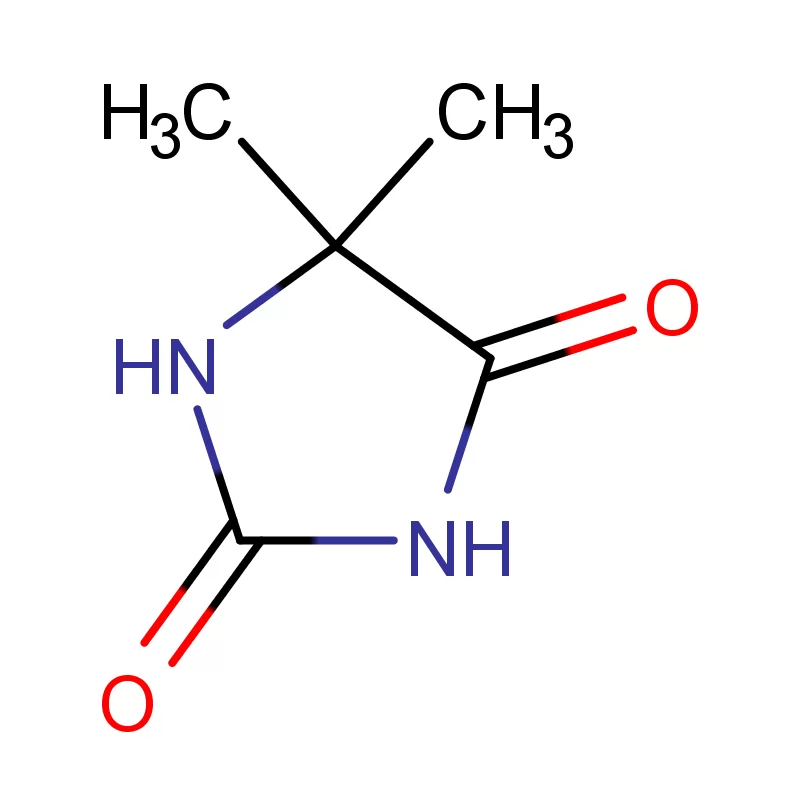
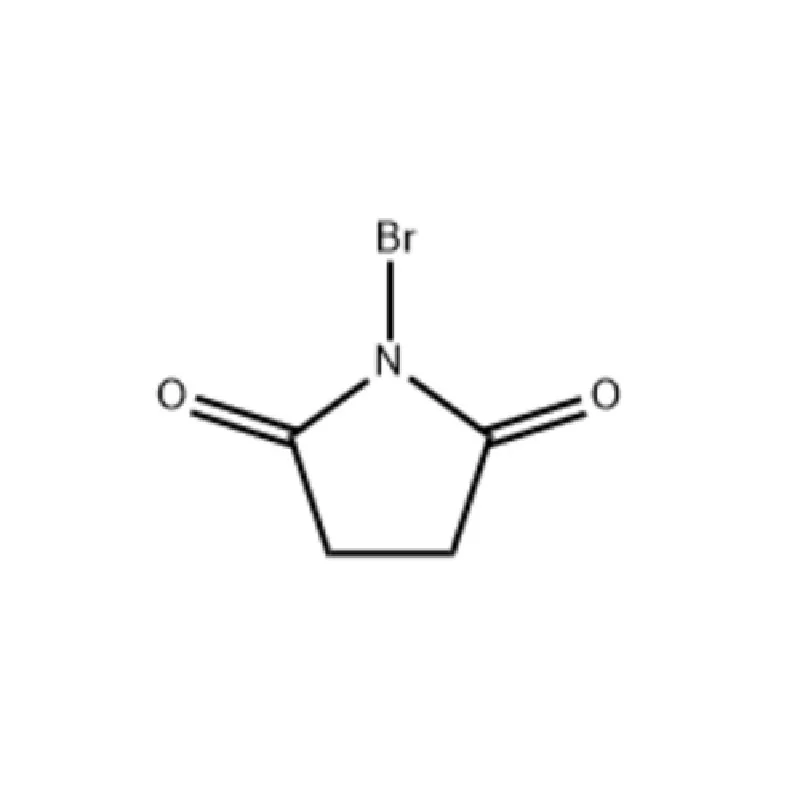
The United Kingdom boasts a strong pharmaceutical sector, driven by substantial investment in research and development. However, recent geopolitical factors and supply chain disruptions have presented challenges for sourcing key pharmaceutical intermediates. Brexit has introduced new complexities regarding import/export regulations, requiring manufacturers to diversify their sourcing strategies.
The UK’s stringent regulatory environment, overseen by the Medicines and Healthcare products Regulatory Agency (MHRA), demands consistently high-quality fine chemicals. This emphasis on quality control creates a demand for reliable suppliers with robust quality assurance systems. The sector is also heavily focused on sustainability and green chemistry principles.
Geographically, pharmaceutical manufacturing hubs are concentrated in regions like Cambridge, Oxford, and the North West of England. These clusters benefit from proximity to leading universities and research institutions, fostering innovation and collaboration. 2 Bromothiophene and specialized chemical reagents are essential components in many UK-based research projects.