Tracing the development of pharmaceutical manufacturing from traditional methods to modern practices.
Market Development History
Prior to the 1990s, Zimbabwe had a relatively robust pharmaceutical manufacturing base, producing a significant portion of its essential medicines. This era relied on basic chemical synthesis and formulation techniques. The liberalization of the economy in the 1990s led to increased competition from imports, impacting local production.
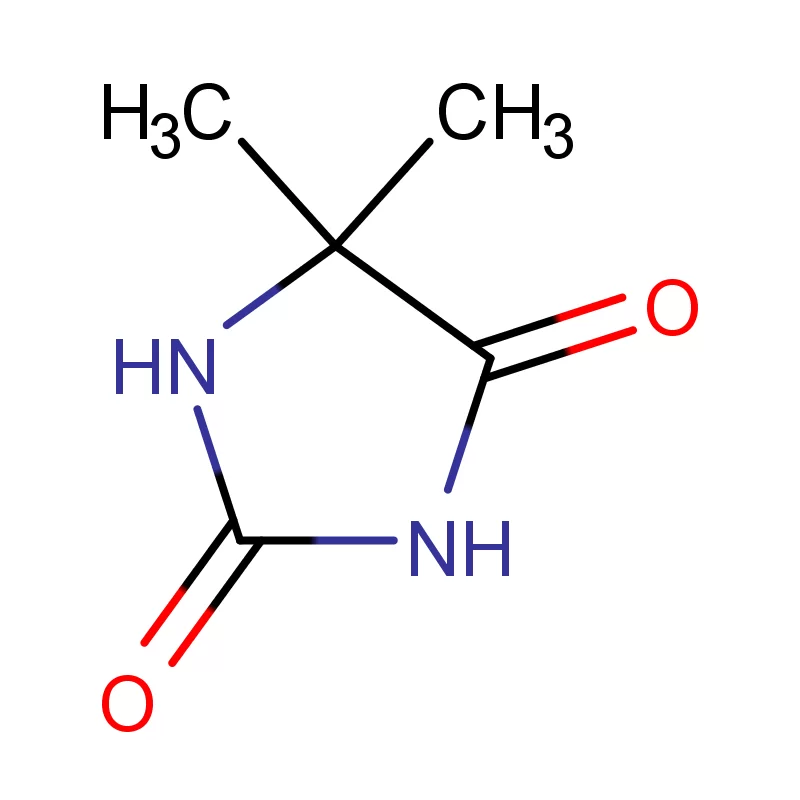
The early 2000s saw a decline in local manufacturing due to economic challenges, including hyperinflation and foreign currency shortages. This period was characterized by a heavy reliance on imported finished products and raw materials like 5 5 Dimethyl Hydantoin. The focus shifted towards distribution and retail.
In recent years, there's been a renewed focus on reviving local manufacturing, driven by government policies promoting import substitution and investment in the pharmaceutical sector. This includes the adoption of modern manufacturing technologies and quality control systems, focusing on industrial water treatment chemicals for maintaining clean manufacturing environments.
Future Development Trends
Advanced Manufacturing Technologies
The future will see an increased adoption of continuous manufacturing, automation, and data analytics to improve efficiency and reduce costs. These technologies enable precise control over the production process, enhancing product quality and reducing waste.
Focus on API Production
There's a growing trend towards local production of active pharmaceutical ingredients (APIs) to reduce reliance on imports and enhance self-sufficiency. This requires significant investment in research and development and specialized manufacturing facilities.
Sustainable Manufacturing Practices
The pharmaceutical industry is increasingly focused on sustainability, including reducing environmental impact and minimizing waste. This involves adopting green chemistry principles, optimizing resource utilization, and implementing responsible waste management practices.